
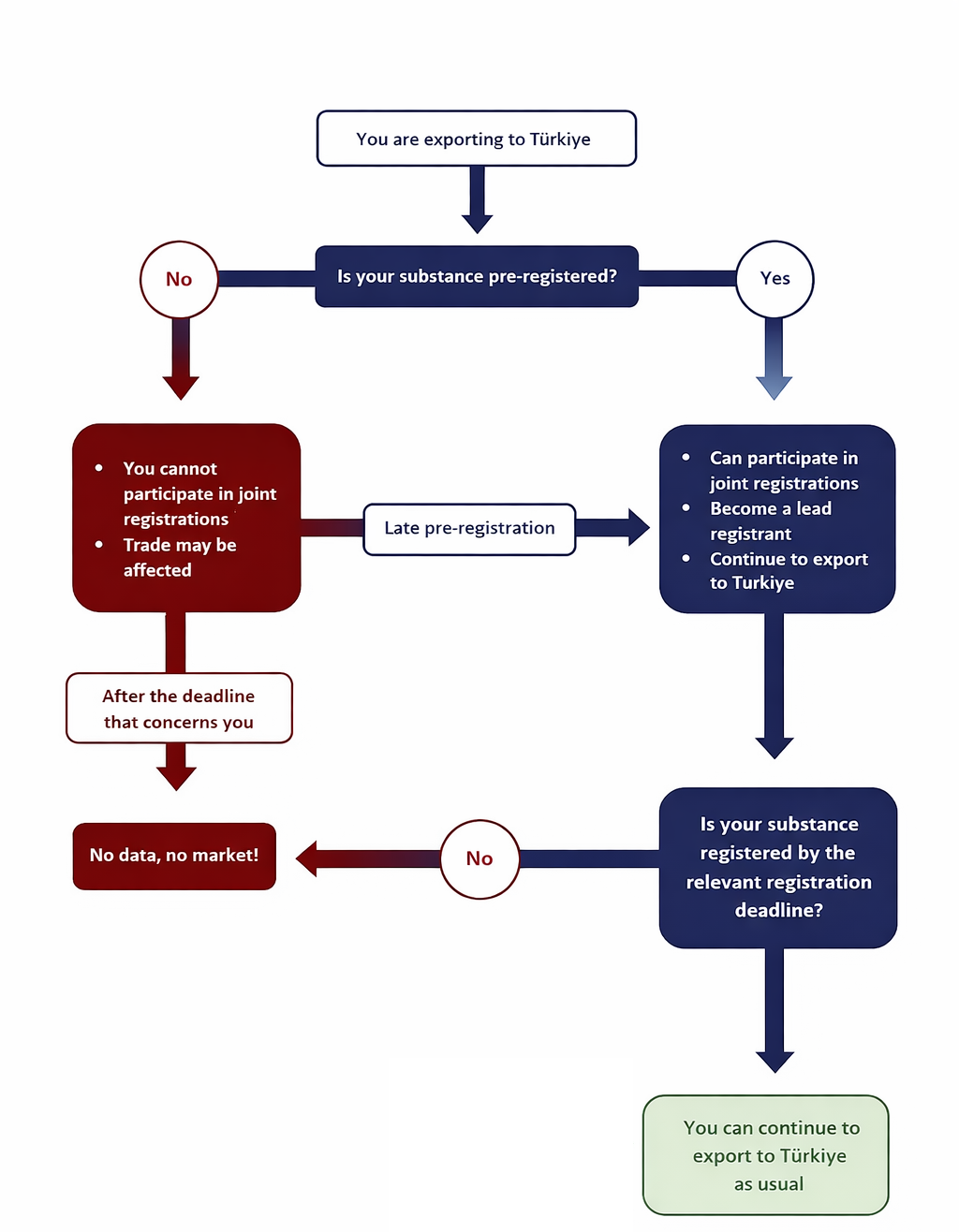
KKDIK came into force as a result of EU Adaptation policy. The first phase of pre-registration ended on December 31st, 2020. The registration phase then officially started the next day (January 1st, 2021). By complying with KKDIK, exporters can ensure their access to the Turkish Market. Exporters of those who wish to enter into the Turkish market but missed the pre-registration should pre-register as soon as possible to be able to benefit from the advantages of joint registration. So far, GPC has successfully helped its clients to pre-register over 3500 substances.
KKDIK (aka Türkiye REACH) came into force on 23rd of June 2017 and has replaced three existing regulations on chemicals and mixtures with the purpose of regulating the chemical inventory, enforcing secure usage, and risk management on hazardous substances, and promoting alternatives to hazardous substances or procedures.
The regulation provides the means of registration, evaluation, authorization, and restrictions of chemicals and safeguarding human health and the environment. It also encourages using alternative methods for evaluating the hazard of substances such as decreasing the number of animal testing and encouraging innovation so that the competitiveness of the Turkish chemical industry will be enhanced.
KKDIK Timeline consist of 3 main phases:
It is strongly recommended for exporters to Turkey to submit late pre-registrations (LPR) as soon as possible to secure business if not done already before 31st December 2020. Pre-registration carries crucial importance to be able to actively participate in SIEF activities for joint registrations.
KKDIK allows only pre-registered substances to be placed in the Turkish market from the 1st of January 2021 and onwards until the relevant deadline of registration. Pre-registration is a simple activity without major compliance requirements that also allows GPC to represent your best interest within the SIEFs.
KKDIK is a comprehensive regulation. However, some substances are exempted from registration either because they are covered by specific regulations such as radioactive waste or regarded as less risky such as polymers. The registration process is not the same for every substance for instance under some conditions, registration of on-site or transported isolated intermediates is relatively easy. You can find information about exempted substances on our website or you can e-mail us at Turkey-REACH@gpcregulatory.com to find out more about the registration process and exempted substances.
Registration requirements change depending on exported tonnage of substance. When the tonnage increases, additional information is needed. For instance, when the tonnage of substance is 10-100 tons per annum, chemical safety report should be provided. Therefore, the best and easiest action would be e-mailing us your substance details and we will provide you your customized complete obligations under Turkey REACH.
The aim of joint registration is to decrease the cost of registration by splitting the fee etc. and reduce the number of animal testing. Lead registrants, who are appointed by the SIEF members for each substance, prepare and submit the joint registration dossier on behalf of the SIEF. GPC can undertake the lead registrant role upon your request. Having managed approximately 1200 registration dossiers including 400 lead registrations in EU REACH, GPC is the holder of data for many substances and thus, we offer you the advantage of having a much faster & cost-effective registration process for the SIEFs that we are the lead of.
News

The Turkish Ministry of Environment, Urbanization and Climate Change (MoEUCC) has issued an announcement requiring all chemical manufacturers and importers operating in Turkey to submit their transitional registration dossiers under the...
Read More_1024x768.webp)
The Turkish Ministry of Agriculture and Forestry has published amendments to the Turkish Food Codex Regulation on Specifications for Food Additives, bringing national legislation into closer alignment with European Union standards....
Read MoreIf you want to access the GHS report, please Register here in GPC Intelligence Portal click here
