
The local government issued Resolution 192/2019, which created a national list of existing, restricted, and prohibited chemical substances, and is continuously being incorporated. Moreover, Decree 504/2019 was closely published, implementing an interministerial group responsible for coordinating different governmental areas to guarantee national management of chemical substances, fulfilling the national and international commitments assumed.
A significant development in Argentina's chemicals regulatory framework is the draft National Law for Risk Management of Chemical Substances (Bill 4339-D-2019), which aims to establish a National Inventory of Chemical Substances (INSQ). This bill, which is currently awaiting approval in Congress, aims to harmonise the regulation of chemical substances at the national level by creating a comprehensive inventory and defining mechanisms for risk assessment and management. The bill includes specific exemptions, including radioactive substances, non-isolated intermediates and substances used in food and medicine. The draft law of national chemical legislation was presented to public discussion in September 2019, right before the country’s election in October. In the end, the change in government affected further progress and the draft was withdrawn by Argentina’s executive power. However, despite expectations for re-introduction in 2021, the law has not yet been approved or fully implemented. Some updates in chemical management have occurred, such as the creation of a national list of regulated chemicals through Resolution 504/2022 and the implementation of the Decree 593/19, which created the National Registry of Chemical Precursors (Registro Nacional de Precursores Químicos). This was further expanded by Decree 606/23, which introduced List I of controlled chemical substances. However, these decrees lack the comprehensive and enduring impact that a national legislation would provide.
|
Country |
Argentina |
|
National Regulation |
|
|
Status |
Draft |
|
Regulation name |
Draft National Law for the management of the risk of chemical substances Anteproyecto de Ley Nacional para la gestión del riesgo de sustancias químicas - link |
|
Objective |
Establish a harmonized inventory at the national level for all chemical substances produced or introduced into the country, as well as the definition or strengthening of risk assessment and management mechanisms |
|
Scope |
Imported or manufactured substances, including mixtures |
|
Further substance lists |
There is no further list of substances of concern on the current draft. A separate list is available here. |
|
Exemption |
Substances intended for research, radioactive, residues, on transit or warehouse, unintended, used in medicines or food additives, and natural substances |
|
Affected stakeholders |
Importers and producers when dealing with amounts ≥ 1 ton per year |
|
Requirements |
It is required to provide the following information:
|
|
Non-compliance |
Sanctions’ modalities:
|
|
Comments |
A new draft version is expected to come out in 2021 defining:
The new draft is to be presented to Congress later in October 2021, after national elections |
Adoption of GHS
Argentina officially adopted GHS 5th version in 2017 through Resolution 801/2015, which was amended twice by Resolution 3.359/2015 and Resolution 155/2016. Altogether, GHS became mandatory for substances on January 1st, 2017, and for mixture from June 1st, 2017. In Argentina, the GHS is supplemented by the Argentine Institute of Standardization and Certification (IRAM) norms 41.400/2013 (SDS) and IRAM 41.401/2020 (Labels).
|
SDS / GHS Regulation |
|
|
Status |
In place (GHS 5th version) |
|
Regulation name |
Legislations:
Standards:
|
|
Implementation date |
Workplace:
Transport:
|
|
Language |
Spanish |
Authorties
National Service of Agricultural Food Health and Quality (SENASA), Argentina's pesticide management body, is in charge of pesticide registration oversight. Pesticides are inspected and tested by the national food inspection and quarantine bureau. Argentina's pesticide registration system needs a single registration licence for each pesticide. Furthermore, if a company wants to register a formulation, it must first register technical material. The Plant Protection Act (Law No. 4975) is the principal pesticide regulation.
Overview
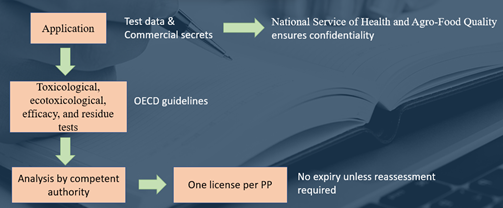
A phytosanitary product must be registered in the National Register of Plant Therapeutics, either experimentally or commercially, in accordance with the provisions of Decree No 3489/58 and Decree No 5769/59, and in accordance with the Manual of Procedures, Criteria, and Scope for the Registration of Phytosanitary Products in the Argentine Republic, approved by Resolution SAGPYA No 350/99. The Fifth Edition of the MANUAL ON THE DEVELOPMENT AND USE OF FAO SPECIFICATIONS FOR PRODUCTS INTENDED FOR THE PROTECTION OF PLANTS is adopted for Argentina in the Resolution. This is a method of analysing enough scientific evidence to show that a product is useful for its intended purpose and poses no unreasonable dangers to human health or the environment. For product analysis, information on physical, chemical, toxicological, ecotoxicological, and waste properties must come from tests or studies conducted on phytosanitary products to be registered or their equivalents, according to internationally recognised protocols, such as the OECD Guidelines on acute oral toxicity, acute dermal toxicity, inhalation classification, dermal irritation, eye irritation, and skin sensitization.
A phytosanitary product presented to SENASA by a registrant that meets all the requirements set forth in the aforementioned Resolution No 350/99 receives a Certificate of Use and Marketing, allowing the product to be used for all of the purposes for which it was registered, as well as allowing the registrant to market it throughout the National Territory.
Who can Register?
A legal entity might be a person or a firm registered with the Department of Trade and Industry under the Company Act.
Pesticides are only allowed to be registered if they are approved to be effective, safe, and of high quality.
For oversea manufacturers, they should appoint an Authorized Representative (AR).
Registration Process
To register pesticide it takes 8 days to complete the treatment.
Physical-chemical property determinations in phytosanitary products, phytotoxicity of oils and adjuvants, seed staining tests, and active ingredient identification and quantification assays.
- Typical turnaround time: 8 business days
- Extremely urgent: 5 business days
- Phytotoxicity determination: 15 business days
7-day processing time
Inorganic Contaminant Determination
Typical Time:
7 business days for one analysis
14 business days for two or three analyses
18 business days for more than three analyses
Urgent:
5 business days for one test
Two or three tests: 7 business days
More than three tests: 14 business days
Registration Process
Compliance Strategies
GPC can help you with:
If you want to access the GHS report, please Register here in GPC Intelligence Portal click here
