
Overview
China's State Council published the final version of the COsmetic Supervision and Administration Regulation (CSAR) and this came into force on 1 January 2021 and replaced the Cosmetic Hygiene Supervision REgulations. CSAR regulates all cosmetics and cosmetic raw materials in China. All the manufacturers, importers and exporters must ensure that their cosmetic products and raw ingredients meet the compliance obligations under CSAR before producing, importing, and exporting business.
Substances are divided into two categories:
Based on different types of substances, different submission procedures (record filing and registration) apply.
Cosmetic Existing Raw Materials
Cosmetic raw material is defined as natural or synthesis ingredients which are used in making cosmetic products. According to the requirements, existing ingredients need to do record filing submission, while new ingredients must do a formal registration to produce or place in China. The distinguished point for checking the procedure for ingredients Is to check if it falls within the cosmetic inventory.
IECIC stands for Inventory of Existing Cosmetic Ingredients in China. Currently, it contains 8972 existing cosmetic ingredients. In addition to the main inventory, there are seven cosmetic inventory lists that can be used in parallel with IECIC. If ingredients are found on the inventory, then it only needs to submit record filing.
|
Inventory lists |
Number listed |
Obligation under list |
|
List of banned ingredients in cosmetics (2021) |
1284 |
Not allow to use |
|
List of banned plant (animal) ingredients in cosmetics (2021) |
109 |
Not allow to use |
|
List of restricted substances in cosmetics (2021) |
47 |
Meet certain requirements |
|
List of preservatives allowed in cosmetics (2015) |
51 |
Allow to use |
|
List of sunscreen agents allowed in cosmetics |
27 |
Allow to use |
|
List of colorants allowed in cosmetics (2015) |
157 |
Allow to use |
|
List of hair dyes allowed in cosmetics (2015) |
75 |
Allow to use |
|
IECIC (2021) |
8972 |
Register / Record Filing |
New Cosmetic Raw Materials
New raw ingredients are defined as cosmetic ingredients that are not listed in the IECIC inventory.
Applicants need to submit the following documents to comply with CSAR:
The application may take up to 4 months before a decision is made. The technical committee will need around 3 months to undertake the evaluation work.
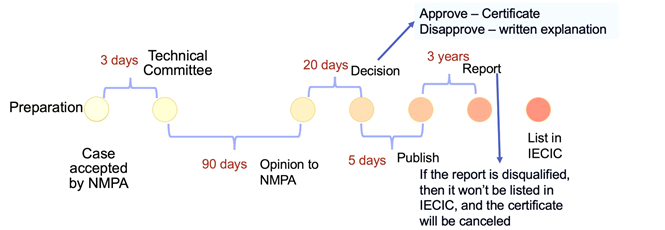
Cosmetic Product Registration
The registration timeline of cosmetic products is the same as cosmetic ingredients registration. But the submission documents are different. For cosmetic products registration, applicants need to prepare:
Label Requirements
For imported cosmetics, they must be labeled in Chinese and contain the following information:
Annual Reporting
CSAR also introduces the annual reporting requirements. Applicants should submit the annual report of cosmetics from January 1 to March 31 each year.
If you want to access the GHS report, please Register here in GPC Intelligence Portal click here
